Introduction
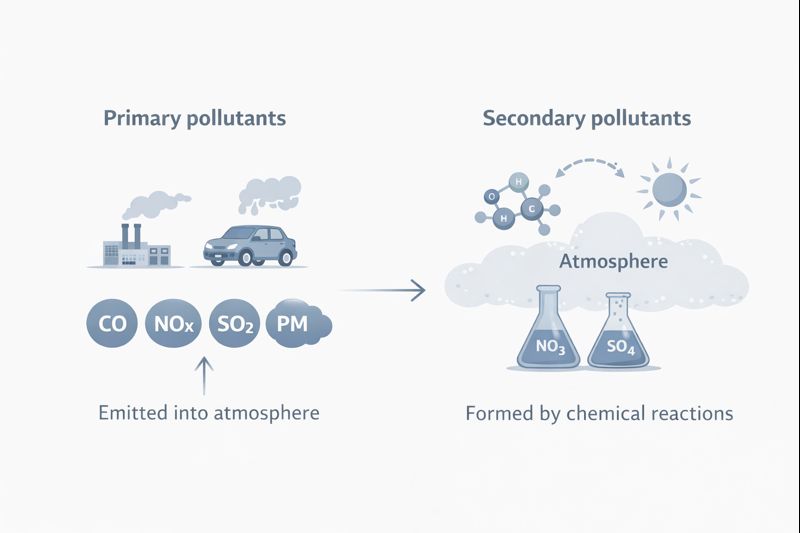
Primary vs secondary pollutants are a fundamental concept in air pollution science. It explains how pollutants enter the atmosphere and how they form in the air. Some pollutants are emitted directly from sources such as vehicles, industries, and power plants, while others develop later through chemical reactions between gases already present in the atmosphere.
Understanding this distinction helps scientists interpret air-quality data, identify pollution sources, and design effective pollution-control strategies. It also explains why pollution levels do not always correspond directly to visible emission sources.
In rapidly urbanizing countries like India—where emissions from transport, industry, construction, and agriculture interact with atmospheric chemistry—distinguishing between primary and secondary pollutants is essential for understanding how pollution episodes develop.
Primary vs Secondary Pollutants: Key Differences
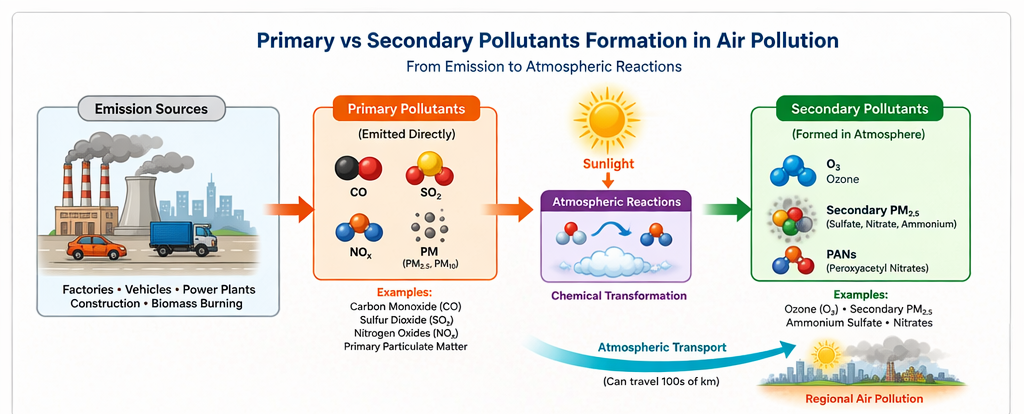
Primary pollutants are air pollutants that are emitted directly into the atmosphere from identifiable sources, such as vehicles, power plants, and industrial facilities.
Secondary pollutants do not enter the atmosphere directly. Instead, they form later through chemical reactions between primary pollutants and atmospheric components such as sunlight, oxygen, or water vapor.
This difference explains why some pollutants appear close to emission sources, while others develop later and spread across wider regions.
Key Differences Between Primary and Secondary Pollutants
| Feature | Primary Pollutants | Secondary Pollutants |
|---|---|---|
| Formation | Emitted directly from sources | Formed through atmospheric chemical reactions |
| Sources | Vehicles, power plants, industries | Reactions between precursor gases |
| Examples | PM₂.₅, CO, SO₂, NOₓ | Ozone, sulfate particles, nitrate particles |
| Spatial pattern | Highest near emission sources | Can spread across large regions |
| Formation time | Immediate | Hours to days after emission |
Examples of Primary and Secondary Pollutants
Common primary pollutants include:
- Particulate matter (PM₂.₅ and PM₁₀)
- Sulfur dioxide (SO₂)
- Nitrogen oxides (NOₓ)
- Carbon monoxide (CO)
Common secondary pollutants include:
- Ground-level ozone (O₃)
- Sulfate particles
- Nitrate particles
- Photochemical smog
What Are Primary Air Pollutants?
Primary air pollutants are substances released directly into the atmosphere from identifiable emission sources.
Their presence in the air is directly linked to those sources.
Major Sources in India
In Indian cities, primary pollutants are commonly associated with:
- Vehicular emissions
- Coal-based thermal power plants
- Construction and road dust
- Industrial operations
- Biomass and crop-residue burning
These emission sources represent some of the major sources of air pollution in Indian cities. In many Indian urban regions, high traffic density and coal-based power generation make these sources significant contributors to ambient pollution levels.
Primary pollutants are measured directly at air quality monitoring stations in India, where instruments continuously record pollutant concentrations. Although they may disperse or react after release, their classification depends solely on how they enter the atmosphere.
What Are Secondary Air Pollutants?
Secondary air pollutants are not emitted directly.
Instead, they form in the atmosphere after primary pollutants undergo chemical reactions.
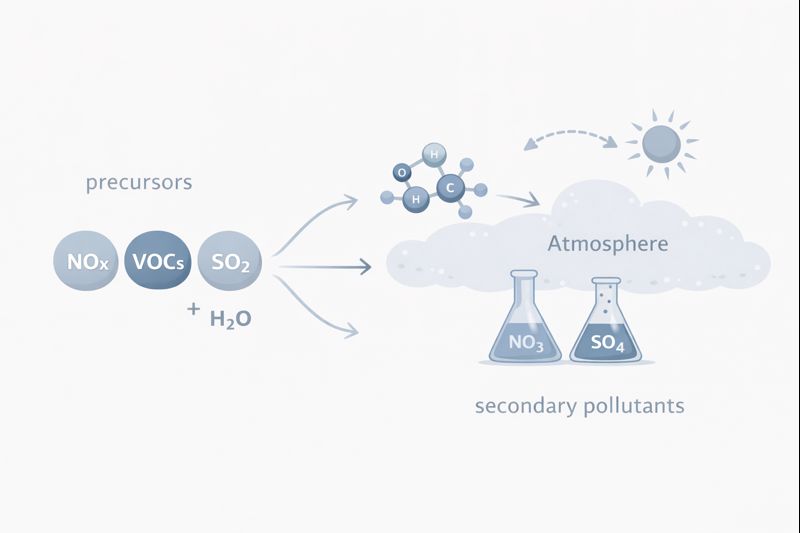
These reactions often involve:
- Nitrogen oxides (NOₓ)
- Sulfur dioxide (SO₂)
- Ammonia (NH₃)
- Volatile organic compounds (VOCs)
Formation Processes in the Atmosphere
Secondary pollutants form through complex chemical reactions occurring in the atmosphere. These reactions are often driven by sunlight, atmospheric oxidants, and interactions between gases released from human activities.
One common example is the formation of ground-level ozone. Nitrogen oxides (NOₓ) and volatile organic compounds (VOCs) emitted from vehicles and industrial processes react in the presence of sunlight to produce ozone. This process is known as photochemical smog formation and is common in large urban areas.
Similarly, sulfur dioxide emitted from coal combustion can oxidize to form sulfate particles, while nitrogen oxides can transform into nitrate particles. These particles combine with ammonia in the atmosphere to produce ammonium sulfate and ammonium nitrate, both of which contribute significantly to fine particulate matter (PM₂.₅).
Photochemical Smog Formation
Photochemical smog is a mixture of secondary pollutants that forms when sunlight drives reactions between nitrogen oxides (NOₓ) and volatile organic compounds (VOCs). These reactions produce ground-level ozone, aldehydes, and other oxidizing chemicals that contribute to urban air pollution. Photochemical smog is commonly observed in large cities where vehicle emissions and strong sunlight accelerate atmospheric chemical reactions.
How Secondary Pollutants Form
Secondary pollutants form through atmospheric chemical reactions involving gases released by human activities and natural processes.
Two common reaction pathways include:
• Photochemical reactions – Sunlight drives reactions between nitrogen oxides (NOₓ) and volatile organic compounds (VOCs), producing ground-level ozone and other oxidants.
• Gas-to-particle conversion – Sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) oxidize in the atmosphere to form sulfate and nitrate particles, which contribute to fine particulate matter (PM₂.₅).
These processes explain why pollution levels can increase even when emission sources remain constant.
Major Sources of Secondary Pollutant Precursors
Secondary pollutants depend on the presence of precursor gases. These precursor emissions originate from a variety of human and natural sources.
In many Indian cities, major sources include:
• Vehicle emissions producing nitrogen oxides and volatile organic compounds
• Coal-based thermal power plants releasing sulfur dioxide
• Agricultural activities emitting ammonia from fertilizers and livestock waste
• Industrial processes generating various chemical gases
• Crop-residue burning that releases large quantities of reactive gases
When these gases mix in the atmosphere under suitable weather conditions, they react to form secondary pollutants that can significantly increase air pollution levels across large regions.
Common Examples
- Ground-level ozone
- Secondary particulate matter (formed from gaseous precursors)
For example:
- Ground-level ozone forms when nitrogen oxides and volatile organic compounds react in the presence of sunlight.
- Secondary particulate matter forms when gases such as sulfur dioxide, nitrogen oxides, or ammonia chemically transform into fine particles.
Many secondary pollutants form through atmospheric oxidation reactions. For example, sulfur dioxide (SO₂) emitted from coal combustion can oxidize to form sulfate particles, while nitrogen oxides (NOₓ) can transform into nitrate particles. These particles often combine with ammonia (NH₃) in the atmosphere to produce compounds such as ammonium sulfate and ammonium nitrate, which are major components of fine particulate matter (PM₂.₅) in polluted urban environments.
In India, winter smog episodes in cities such as Delhi often involve a significant secondary component. Low wind speeds and temperature inversion conditions allow atmospheric reactions to intensify pollution levels.
Influence of Atmospheric Conditions
The formation and accumulation of secondary pollutants depend heavily on:
- Sunlight intensity
- Temperature
- Humidity
- Wind patterns
- Air mass movement
Because of these factors, secondary pollution levels may rise even when emission sources remain relatively stable.
This explains why pollution episodes can sometimes appear disproportionate to visible emission activity.
A well-known example of secondary pollution occurs during winter smog episodes in Delhi and northern India. During these events, emissions from vehicles, industries, and agricultural burning release large amounts of precursor gases such as nitrogen oxides, sulfur dioxide, and ammonia. Under conditions of low wind speed and temperature inversion, these gases undergo chemical reactions in the atmosphere, forming large quantities of secondary particulate matter that significantly increase PM₂.₅ concentrations.
Secondary Pollution in Indian Cities
Air pollution episodes in Indian metropolitan regions often involve a strong secondary component. Cities such as Delhi frequently experience winter smog events where atmospheric chemistry intensifies pollution levels.
During winter months, temperature inversions trap pollutants close to the ground while weak wind speeds prevent dispersion. At the same time, emissions from vehicles, power plants, construction activities, and crop burning release large quantities of precursor gases. These gases react in the atmosphere to form secondary particulate matter, significantly increasing PM₂.₅ concentrations.
Because secondary pollutants can travel long distances, pollution observed in a city may partly originate from emissions occurring hundreds of kilometers away.
These atmospheric processes explain why pollution levels sometimes remain high even after emission reductions.
Why the Distinction Matters
The distinction between primary and secondary pollutants helps explain why air pollution levels do not always decrease immediately after emission reductions. When direct emission sources are controlled, concentrations of primary pollutants generally decrease. However, secondary pollutants can continue forming through chemical reactions involving precursor gases already present in the atmosphere.
For example, even if direct particulate emissions are reduced, gases such as sulfur dioxide (SO₂), nitrogen oxides (NOₓ), and ammonia (NH₃) may still react to produce secondary particulate matter.
These atmospheric reactions can continue for several hours or even days after pollutants are emitted, depending on weather conditions such as sunlight, temperature, and wind patterns.
These processes are often reflected in daily air-quality indicators such as the Air Quality Index (AQI), which summarizes pollutant concentrations influenced by both emissions and atmospheric reactions.
Can a Pollutant Be Both Primary and Secondary?
In some cases, a pollutant can exist in both primary and secondary forms. This is especially true for particulate matter (PM₂.₅).
Particulate matter may be emitted directly from sources such as vehicle exhaust, construction dust, industrial combustion, and biomass burning. In these situations, the particles are considered primary pollutants because they enter the atmosphere directly.
However, particulate matter can also form secondarily through atmospheric chemical reactions. Gases such as sulfur dioxide (SO₂), nitrogen oxides (NOₓ), and ammonia (NH₃) can react in the atmosphere to produce sulfate, nitrate, and ammonium particles. These newly formed particles become part of fine particulate matter (PM₂.₅).
Because of this dual formation pathway, scientists often describe particulate pollution as having both primary and secondary components, especially during severe pollution episodes in large urban regions.
Interpretation Limits
The primary–secondary distinction is a practical framework for understanding how air pollution forms. However, atmospheric systems are complex.
Some pollutants, such as particulate matter, may be emitted directly while also forming through chemical reactions. For this reason, the classification is used as an analytical tool rather than as a strict boundary.
Why Controlling Secondary Pollution Is Challenging
Reducing primary emissions does not always produce immediate improvements in air quality because secondary pollutants continue forming in the atmosphere.
Even if direct emissions decrease, previously released gases may still react and produce pollutants for several hours or days. In addition, atmospheric transport can move precursor gases across regions, allowing pollution formed in one area to affect air quality in another.
For this reason, air quality management strategies often focus on reducing precursor gases such as nitrogen oxides, sulfur dioxide, and ammonia rather than targeting particulate matter alone.
Frequently Asked Questions
What are primary pollutants?
Primary pollutants are air pollutants that are emitted directly into the atmosphere from identifiable sources. These sources include vehicle exhaust, industrial emissions, coal combustion, construction dust, and biomass burning. Because primary pollutants enter the air directly, their concentration is often highest near emission sources.
What are secondary pollutants?
Secondary pollutants are not emitted directly into the atmosphere. Instead, they form when primary pollutants react chemically with other substances in the air. These reactions often involve sunlight, oxygen, or water vapor. Common secondary pollutants include ground-level ozone and certain forms of fine particulate matter.
What is the main difference between primary and secondary pollutants?
The key difference lies in how they enter the atmosphere. Primary pollutants are released directly from sources such as vehicles, power plants, or industrial facilities. Secondary pollutants form later in the atmosphere through chemical reactions involving primary pollutants and other atmospheric components.
What are examples of primary pollutants?
Common examples of primary pollutants include:
- Particulate matter (PM₂.₅ and PM₁₀)
- Sulfur dioxide (SO₂)
- Nitrogen oxides (NOₓ)
- Carbon monoxide (CO)
- Volatile organic compounds (VOCs)
These pollutants originate from activities such as fossil-fuel combustion, industrial processes, transportation, and biomass burning.
What are examples of secondary pollutants?
Examples of secondary pollutants include:
- Ground-level ozone (O₃)
- Secondary particulate matter
- Sulfate and nitrate particles
- Photochemical smog
These pollutants form through atmospheric chemical reactions involving precursor gases like nitrogen oxides, sulfur dioxide, and volatile organic compounds.
Why is understanding primary and secondary pollutants important?
Understanding the distinction between primary and secondary pollutants helps scientists interpret air quality data and design effective pollution control strategies. It also explains why pollution levels may remain high even after emission reductions, since atmospheric chemical reactions can continue forming pollutants over time.
Can particulate matter be both primary and secondary?
Yes. Particulate matter can exist in both forms. Some particles are emitted directly from sources such as vehicle exhaust, construction dust, and biomass burning. Others form in the atmosphere when gases such as sulfur dioxide, nitrogen oxides, and ammonia react chemically to produce sulfate, nitrate, and ammonium particles.
Conclusion
Understanding primary vs secondary pollutants provides an essential foundation for studying air pollution.
Primary pollutants originate directly from emission sources, while secondary pollutants form through chemical reactions in the atmosphere. These atmospheric processes explain why pollution can spread across regions and why air-quality improvements sometimes occur slowly.
For scientists, policymakers, and the public, recognizing how these pollutants form is critical for interpreting air-quality data and designing effective pollution control strategies in countries like India.
References
- Central Pollution Control Board (CPCB), Government of India. (n.d.). National Air Quality Index (AQI): Technical Framework.
- Central Pollution Control Board (CPCB), Government of India. National Air Quality Monitoring Programme (NAMP): Guidelines and Methodology.
- Ministry of Environment, Forest and Climate Change (MoEFCC), Government of India. (2009). National Ambient Air Quality Standards (NAAQS).
- World Health Organization (WHO). (2021). WHO Global Air Quality Guidelines. Geneva: WHO.
- Seinfeld, J. H., & Pandis, S. N. (2016). Atmospheric Chemistry and Physics: From Air Pollution to Climate Change (3rd ed.). Wiley.